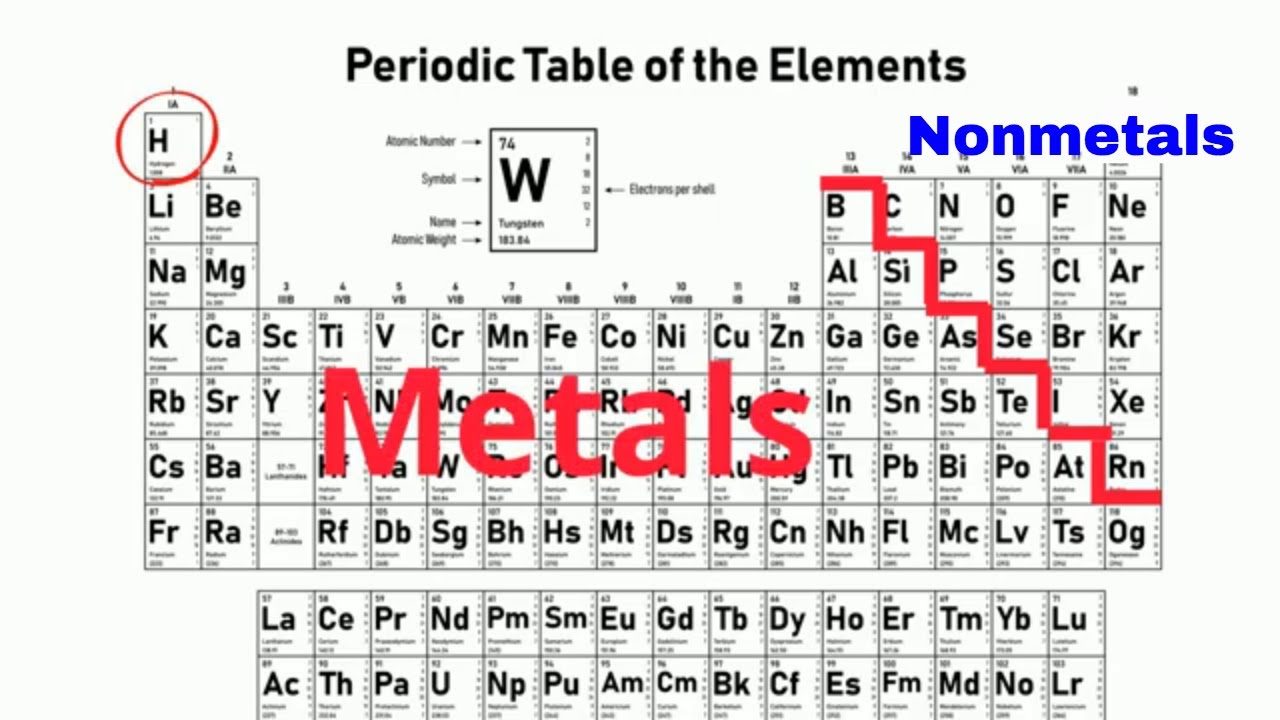
Atomic mass generally increases from left to right across a period because elements are organized by atomic number,meaning that proton and neutron numbers will generally increase from left to right and top to bottom. The horizontal rows of the periodic table are called periods. See also: Alkaline-earth metals Transition elements Column 15, headed by nitrogen, is known as the pnictogens column 16, beginning with oxygen, as the chalcogens column 17, starting with fluorine, as the halogens and column 18, starting with helium, as the noble gases. Columns 13–18 are often discussed along with columns 1 and 2, and, collectively, the elements in these columns are known as the main group or representative elements because these elements are the most abundant on Earth and in the universe overall. Columns 3–12 are filled by the transition metals, which are shiny and good conductors of both heat and electricity. Column 2 contains the alkaline-earth metals, which have higher melting points and are less reactive. In column 1, each alkali metal is soft, relatively low-melting, and highly reactive toward air and water. Elements in each family tend to have similar properties. The modern periodic table is divided into 18 columns called groups or families (see illustration). See also: Alkali metals Atom Atomic number Chemical element Electron Electron configuration Neutron Noble gases Proton Valence The position of elements in the periodic table provides a powerful method of classifying not only the physical properties of elements but also their expected properties in molecules and solids. In contrast to the first column of the periodic table, the last column contains elements with completely filled valence orbitals these are chemically inert elements, meaning they do not react with other atoms, and are called noble gases. Since all spontaneous reactions increase stability, elements react with each other in order to fill their outer (valence) orbital with electrons a filled valence orbital is the most stable configuration for any atom. Valence electrons-electrons in the outer shell of an atom-are important because the number of them determines an element's chemical reactivity, or how the element "behaves" in the presence of other atoms. The first column of the periodic table is known as the alkali metals group (although this column does includes a gas, hydrogen, the very first element in the periodic table), which are elements with just one outer (valence) electron. Atoms are made of subatomic particles, with protons and neutrons comprising the nucleus, and electrons, hovering in orbitals around the nucleus. Each chemical element in the periodic table (see illustration) is organized such that the vertical columns consist of elements with remarkably similar properties. The right side of the periodic table contains the nonmetals (except hydrogen, which is in group 1), including halogens (group 17) and noble gases (group 18).Ī list of all known chemical elements arranged along horizontal rows in increasing atomic number (number of protons) and in vertical columns consisting of elements with remarkably similar properties. The left side of the periodic table includes the alkali (group 1) and alkaline-earth (group 2) metals, followed by the transition metals in the center. Originally developed by the Russian chemist Dimitri Mendeleyev, the periodic table lists every known chemical element organized in horizontal rows (“periods”) by increasing atomic number and in columns (“groups”) by same number of valence electrons and, therefore, similar chemical properties.Įach cell in the periodic table contains a symbol representing the element and its atomic number.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
